Summary
Abstract
Recombinant factor VIIa (NovoSeven®; also known as recombinant activated factor VII or eptacog alfa) is structurally similar to human plasma-derived coagulation factor VIIa, but is manufactured using DNA biotechnology. Recombinant factor VIIa interacts with thrombin-activated platelets to produce a thrombin burst leading to accelerated fibrin clot formation localized to the site of vascular injury. It is approved in many countries for use as an intravenous hemostatic agent in patients with congenital hemophilia with inhibitors, and also for acquired hemophilia, factor VII deficiency, and Glanzmann thrombasthenia in some countries.
Studies have shown it to be effective and generally well tolerated when used intravenously to treat bleeding episodes or provide hemostatic cover during surgery in patients with congenital hemophilia with inhibitors, acquired hemophilia, factor VII deficiency or Glanzmann thrombasthenia. Based on available data, its efficacy in terms of patient-assessed response may be similar to that of activated prothrombin complex concentrate (aPCC), but treatment with a single 270 µg/kg dose of recombinant factor VIIa might reduce the need for rescue therapy compared with aPCC. Recombinant factor VIIa is not immunogenic in patients with hemophilia, does not produce an anamnestic response in hemophilia patients with inhibitors, and has very low thrombogenicity. It is recommended in guidelines as the treatment of choice for bleeds in patients with hemophilia B with high-responding inhibitors and for patients with factor VII deficiency, and is also a first-line therapeutic option for high-responder hemophilia A patients with inhibitors and those with acquired hemophilia. Cost data from pharmacoeconomic analyses support its use in hemophilia patients with inhibitors. Thus, recombinant factor VIIa is a valuable treatment option for patients with these rare, but potentially serious, bleeding disorders.
Pharmacologie Properties
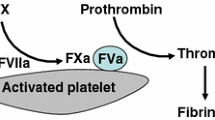
Recombinant factor VIIa is a recombinant protein, similar in structure to coagulation factor VIIa derived from human plasma, but manufactured without using material of human origin. It has effects on both tissue factor-dependent and -independent coagulation. In particular, at pharmacologic concentrations, its main effect is to enhance thrombin generation on the surface of activated platelets, even in the absence of factors VIII and IX, which are deficient in patients with congenital hemophilia A and B, respectively. The thrombin induced by recombinant factor VIIa enhances platelet activation, fibrin formation, and inhibition of fibrinolysis. The effects of recombinant factor VIIa are localized to the site of vascular injury and it does not appear to enhance systemic activation of coagulation. Thrombotic events have been reported rarely. Antibodies to recombinant factor VIIa generally do not develop in patients with hemophilia, although there have been a few reports in patients with factor VII deficiency.
Following a single intravenous dose of recombinant factor VIIa in patients with hemophilia, the area under the plasma concentration-time curve generally increased in a dose-dependent fashion. Volume of distribution at steady state, clearance, elimination half-life, and mean residence time were generally independent of dose in patients with hemophilia. Values for clearance were higher in pediatrie patients than in adults with hemophilia, and were also higher in patients with factor VII deficiency than in adults with hemophilia.
Therapeutic Efficacy
In addition to a number of small controlled trials, therapeutic efficacy data for recombinant factor VIIa come from noncomparative studies, and compassionate- or emergency-use programs. The most widely used dosage in these studies was 90 µ-g/kg every 2–4 hours in patients with congenital hemophilia with inhibitors, acquired hemophilia or Glanzmann thrombasthenia, and 15–30 µ-g/kg in patients with congenital factor VII deficiency. Hemostatic efficacy was generally evaluated using a global assessment by the patient or the physician.
Recombinant factor VIIa was effective at controlling mild to moderate bleeding episodes in patients with congenital hemophilia with inhibitors during home treatment in noncomparative studies, with efficacy rates of up to 93% after ≈2 doses. Early treatment was more likely to be associated with a successful outcome than late treatment.
In double-blind or open-label comparative trials in hemophilia patients with inhibitors experiencing hemarth-roses, the effectiveness of a single dose of recombinant factor VIIa 270 µg/kg was similar to that of a standard regimen of 90 µg/kg every 3 hours. A single 270 µg/kg dose achieved successful hemostasis in >90% of patients with mild to moderate bleeding episodes after 9 hours without the need for additional hemostatic medication.
Two open-label, crossover studies compared recombinant factor VIIa with aPCC in hemophilia patients with inhibitors. One study failed to demonstrate equivalency between the two treatments, possibly due to inadequate patient/bleed numbers. In the other study, patient-assessed treatment response did not differ significantly between recombinant factor VIIa (single dose of 270 µg/kg or standard regimen of three doses of 90 µg/kg at 3-hour intervals) and aPCC 75 U/kg; however, fewer patients required ‘rescue’ therapy with additional hemostatic agents after a single 270 µ-g/kg dose of recombinant factor VIIa than after aPCC.
Based on data largely from the compassionate-use program, recombinant factor VIIa was effective in the treatment of major nonsurgical bleeds (e.g. life- or limb-threatening bleeds), and at producing and maintaining hemostasis during surgery, in patients with hemophilia with inhibitors. In a double-blind controlled trial in hemophilia patients with inhibitors undergoing surgical procedures, satisfactory hemostasis was achieved in 13 out of 14 patients (93%) treated with a standard regimen of recombinant factor VIIa.
Data from a limited number of patients with acquired hemophilia, factor VII deficiency or Glanzmann thrombasthenia (mostly from the compassionate-use programs or patient registries) indicated that recombinant factor VIIa was also effective at treating surgical and nonsurgical bleeding in these patient groups. Efficacy rates were generally consistent with those seen in patients with congenital hemophilia.
Tolerability
Recombinant factor VIIa is generally well tolerated and adverse events do not appear to be dose-related. Non-serious adverse events include nausea, fever, injection-site pain, skin rash and increased values for ALT, alkaline phosphatase and lactate dehydrogenase. The overall incidence of serious adverse events with recombinant factor VIIa is <1%. Serious events have included thrombotic events (e.g. myocardial infarction, stroke, pulmonary embolism, deep vein thrombosis, disseminated intravascular coagulation); however, they have generally occurred in patients with predisposing risk factors or in patients without hemophilia being treated for non-approved conditions. Recombinant factor VIIa has no risk for transmission of human pathogens, is not immunogenic, and does not induce an anamnestic response in patients with inhibitors.
Pharmacoeconomic Considerations
On-demand treatment with recombinant factor VIIa was associated with improvements in health-related quality of life relative to plasma-derived agents in a cost-utility study in hemophilia patients with inhibitors. Modelled cost analyses in this patient population found that on-demand treatment of mild to moderate bleeds with recombinant factor VIIa was cost neutral or cost saving relative to aPCC. Modelled cost analyses also showed that orthopedic surgery using recombinant factor VIIa to maintain hemostasis was generally cost saving over the medium to long term relative to not having surgery. Modelled analyses were performed from the healthcare payer perspective.





Similar content being viewed by others
Notes
The use of trade names is for product identification purposes only and does not imply endorsement.
References
Mannucci PM, Tuddenham EGD. The hemophilias: from royal genes to gene therapy. New Eng J Med 2001 Jun 7; 344(23): 1773–9
Bolton-Maggs PHB, Perry DJ, Chalmers EA, et al. The rare coagulation disorders— review with guidelines for management from the United Kingdom Haemophilia Centre Doctors’ Organisation. Haemophilia 2004 Sep; 10(5): 593–628
D’Andrea G, Margaglione M, on behalf of the Glanzmann’s Thrombas-thenialtalian Team (GLATIT). Glanzmann’s thrombasthenia: modulation of clinical phenotype by α2C807T gene polymorphism. Haematologica 2003; 88(12): 1378–82
Perry DJ. Factor VII deficiency. Blood Coagul Fibrinolysis 2003; 14Suppl. 1: S47–54
Poon MC, D’Oiron R, Von Depka M, et al. Prophylactic and therapeutic recombinant factor VIIa administration to patients with Glanzmann’s thrombasthenia: results of an international survey. J Thromb Haemost 2004 Jul; 2(7): 1096–103
Bolton-Maggs PHB, Pasi KJ. Haemophilias A and B. Lancet 2003 May 24; 361: 1801–9
United Kingdom Haemophilia Centre Doctors’ Organisation (UKHCDO). Guidelines on the selection and use of therapeutic products to treat haemophilia and other hereditary bleeding disorders. Haemophilia 2003 Jan; 9(1): 1–23
Green D. Complications associated with the treatment of haemophiliacs with inhibitors. Haemophilia 1999 Sep; 5Suppl. 3: 11–7
Negrier C, Lienhart A. Overall experience with NovoSeven®. Blood Coagul Fibrinolysis 2000 Apr; 11Suppl. 1: S19–24
Siddiqui MA, Scott LJ. Recombinant factor VIIa (Eptacog Alfa): a review of its use in congenital or acquired haemophilia and other congenital bleeding disorders. Drugs 2005; 65(8): 1161–77
Novo Nordisk Limited. Novoseven 1.2mg (60 KIU) 2.4mg (120 KIU) and 4.8mg (240 KIU) powder and solvent for solution for injection [online]. Available from URL: http://www.emc.medicines.org.uk [Accessed 2007 Dec 12]
Novo Nordisk Inc. NovoSeven® Coagulation Factor VIIa (recombinant). US prescribing information. [online]. Available from URL: http://www.novoseven.com [Accessed 2007 Dec 14]
Hedner U. Mechanism of action of factor VIIa in the treatment of coagulopathies. Semin Thromb Hemost 2006 Apr; 32Suppl. 1: 77–85
Poon MC. The evidence for the use of recombinant human activated factor VII in the treatment of bleeding patients with quantitative and qualitative platelet disorders. Transfus Med Rev 2007 Jul; 21(3): 223–36
Hedner U. Mechanism of action of recombinant activated factor VII: an update. Semin Hematol 2006 Jan; 43(1 Suppl. 1): S105–7
Butenas S, Brummel KE, Bouchard BA, et al. How factor VIIa works in hemophilia. J Thromb Haemost 2003 Jun; 1(6): 1158–60
Gallistl S, Cvirn G, Muntean W. Recombinant factor VIIa does not induce hypercoagulability in vitro. Thromb Haemost 1999; 81(2): 245–9
Allen GA, Hoffman M, Roberts HR, et al. Manipulation of prothrombin concentration improves response to high-dose factor VIIa in a cell-based model of haemophilia. Br J Haematol 2006 Aug; 134(3): 314–9
Butenas S, Brummel KE, Branda RF. Mechanism of factor VIIa-dependent coagulation in hemophilia blood. Blood 2002 Feb 1; 99(3): 923–30
van’t Veer C, Golden NJ, Mann KG. Inhibition of thrombin generation by the zymogen factor VII: implications for the treatment of hemophilia A by factor VIIa. Blood 2000 Feb 15; 95(4): 1330–5
Butenas S, Brummel KE, Paradis SG, et al. Influence of factor VIIa and phospholipids on coagulation in “acquired” hemophilia. Arterioscler Thromb Vasc Biol 2003 Jan 1; 23(1): 123–9
Hoffman M, Monroe DM, Roberts HR. Activated factor VII activates factors IX and X on the surface of activated platelets: thoughts on the mechanism of action of high-dose activated factor VII. Blood Coagul Fibrinolysis 1998; 9Suppl. 1: S61–5
Monroe DM, Hoffman M, Oliver JA, et al. A possible mechanism of action of activated factor VII independent of tissue factor. Blood Coagul Fibrinolysis 1998; 9Suppl. 1: S15–20
Lisman T, Adelmeijer J, Cauwenberghs S, et al. Recombinant factor VIIa enhances platelet adhesion and activation under flow conditions at normal and reduced platelet count. J Thromb Haemost 2005; 3: 742–51
Wolberg AS, Allen GA, Monroe DM, et al. High dose factor VIIa improves clot structure and stability in a model of haemophilia B. Br J Haematol 2005 Dec; 131(5): 645–55
He S, Blombäck M, Jacobsson Ekman G, et al. The role of recombinant factor VIIa (FVIIa) in fibrin structure in the absence of FVIII/FIX. J Thromb Haemost 2003; 1: 1215–9
Lisman T, Mosnier LO, Lambert T, et al. Inhibition of fibrinolysis by recombinant factor VIIa in plasma from patients with severe hemophilia A. Blood 2002 Jan 1; 99(1): 175–9
Antovic JP, Antovic A. Does recombinant factor VIIa, apart from overall hemostasis, regulate TAFI dependent fibrinolysis? In vitro analysis using overall hemostasis potential (OHP) assay. Thromb Haemost 2003 Oct; 90(4): 620–7
Livnat T, Zivelin A, Martinowitz U, et al. Prerequisites for recombinant factor VIIa-induced thrombin generation in plasmas deficient in factors VIII, IX or XI. J Thromb Haemost 2006 Jan; 4(1): 192–200
Galan AM, Tonda R, Pino M, et al. Increased local procoagulant action: a mechanism contributing to the favorable hemostatic effect of recombinant FVIIa in PLT disorders. Transfusion (Paris) 2003 Jul; 43(7): 885–92
Lisman T, Adelmeijer J, Heijnen HFG, et al. Recombinant factor VIIa restores aggregation of αIIbβ3-deficient platelets via tissue factor-independent fibrin generation. Blood 2004 Mar 1; 103(5): 1720–7
Macik BG, Lindley CM, Lusher J, et al. Safety and initial clinical efficacy of three dose levels of recombinant activated factor VII (rFVIIa): results of a phase I study. Blood Coagul Fibrinolysis 1993; 4: 521–7
Kavakli K, Makris M, Zulfikar B, et al. Home treatment of haemarthroses using a single dose regimen of recombinant activated factor VII in patients with haemophilia and inhibitors: a multi-centre, randomised, double-blind, crossover trial. Thromb Haemost 2006 Apr; 95(4): 600–5
Shapiro AD, Gilchrist GS, Hoots WK, et al. Prospective, randomised trial of two doses of rFVIIa (NovoSeven) in haemophilia patients with inhibitors undergoing surgery. Thromb Haemost 1998; 80: 773–8
Pruthi RK, Mathew P, Valentino LA, et al. Haemostatic efficacy and safety of bolus and continuous infusion of recombinant factor VIIa are comparable in haemophilia patients with inhibitors undergoing major surgery: results from an open-label, randomized, multicenter trial. Thromb Haemost 2007 Oct; 98(4): 726–32
Nicolaisen EM. Antigenicity of activated recombinant factor VII followed through nine years of clinical experience. Blood Coagul Fibrinolysis 1998 Mar; 9Suppl. 1: S119–23
Astermark J, Ekman M, Berntorp E. Antibodies to factor VIIa in patients with haemophilia and high-responding inhibitors. Br J Haematol 2002 Nov; 119(2): 342–7
Lusher JM, Roberts HR, Davignon G, et al. A randomized, double-blind comparison of two dosage levels of recombinant factor VIIa in the treatment of joint, muscle and mucocutaneous haemorrhages in persons with haemophilia A and B, with and without inhibitors. rFVIIa Study Group. Haemophilia 1998; 4: 790–8
Mariani G, Testa MG, Di Paolantonio T, et al. Use of recombinant, activated factor VII in the treatment of congenital factor VII deficiencies. Vox Sang 1999; 77(3): 131–6
Ingerslev J, Christiansen K, Sørensen B, for the International Registry on Factor VII Deficiency (IRF7) Steering Committee. Inhibitor to factor VII in severe factor VII deficiency: detection and course of the inhibitory response [letter]. J Thromb Haemost 2005; 3(4): 799–800
Johannessen M, Andreasen RB, Nordfang O. Decline of factor VIII and factor IX inhibitors during long-term treatment with NovoSeven®. Blood Coagul Fibrinolysis 2000 Apr; 11(3): 239–42
Hay CRM, Brown S, Collins PW, et al. The diagnosis and management of factor VIII and IX inhibitors: a guideline from the United Kingdom Haemophilia Centre Doctors Organisation. Br J Haematol 2006 Jun; 133(6): 591–605
Fridberg MJ, Hedner U, Roberts HR, et al. A study of the pharmacokinetics and safety of recombinant activated factor VII in healthy Caucasian and Japanese subjects. Blood Coagul Fibrinolysis 2005; 16(4): 259–66
Girard P, Nony P, Erhardtsen E, et al. Population pharmacokinetics of recombinant factor VIIa in volunteers anticoagulated with acenocoumarol. Thromb Haemost 1998; 80: 109–13
Lindley CM, Sawyer WT, Macik BG, et al. Pharmacokinetics and pharmacodynamics of recombinant factor VIIa. Clin Pharmacol Ther 1994 Jun; 55: 638–48
Villar A, Aronis S, Morfini M, et al. Pharmacokinetics of activated recombinant coagulation factor VII (NovoSeven®) in children vs. adults with haemophilia A. Haemophilia 2004 Jul; 10(4): 352–9
Berrettini M, Mariani G, Schiavoni M, et al. Pharmacokinetic evaluation of recombinant, activated factor VII in patients with inherited factor VII deficiency. Haematologica 2001; 86(6): 640–5
Klitgaard T, Nielsen TG. Overview of the human pharmacokinetics of recombinant activated factor VII. Br J Clin Pharmacol 2008 Jan; 65(1): 3–11
Bernstein DE, Jeffers L, Erhardtsen E, et al. Recombinant factor VIIa corrects prothrombin time in cirrhotic patients: a preliminary study. Gastroenterology 1997; 113: 1930–7
Young G, Shafer FE, Rojas P, et al. Single 270 ug kg-1-dose rVIIa vs. standard 90 ug kg-1-dose rVIIa and APCC for home treatment of joint bleeds in haemophilia patients with inhibitors: a randomized comparison. Haemophilia Epub 2007 Dec 10
Santagostino E, Mancuso ME, Rocino A, et al. A prospective randomized trial of high and standard dosages of recombinant factor VIIa for treatment of hemarth-roses in hemophiliacs with inhibitors. J Thromb Haemost 2006 Feb; 4(2): 367–71
Astermark J, Donfield SM, DiMichele DM, et al. A randomized comparison of bypassing agents in hemophilia complicated by an inhibitor: the FEIBA NovoSeven Comparative (FENOC) Study. Blood 2007 Jan 15; 109(2): 546–51
Key NS, Aledort LM, Beardsley D, et al. Home treatment of mild to moderate bleeding episodes using recombinant factor VIIa (Novoseven) in haemophiliacs with inhibitors. Thromb Haemost 1998; 80: 912–8
Ingerslev J, Thykjaer H, Scheibel E. Approaches towards successful home treatment in patients with inhibitors. Eur J Haematol 1998; 61(Suppl. 63): 11–4
Santagostino E, Gringeri A, Mannucci PM. Home treatment with recombinant activated factor VII in patients with factor VIII inhibitors: the advantages of early intervention. Br J Haematol 1999; 104: 22–6
Arkin S, Cooper HA, Hutter JJ, et al. Activated recombinant human coagulation factor VII therapy for intracranial hemorrhage in patients with hemophilia A or B with inhibitors. Haemostasis 1998; 28: 93–8
Arkin S, Blei F, Fetten J, et al. Human coagulation factor FVIIa (recombinant) in the management of limb-threatening bleeds unresponsive to alternative therapies: results from the NovoSeven® emergency-use programme in patients with severe haemophilia or with acquired inhibitors. Blood Coagul Fibrinolysis 2000 Apr; 11(3): 255–9
Bech RM. Recombinant factor VIIa in joint and muscle bleeding episodes. Haemostasis 1996; 26Suppl. 1: 135–8
Lusher JM. Recombinant factor VIIa (NovoSeven®) in the treatment of internal bleeding in patients with factor VIII and IX inhibitors. Haemostasis 1996 Jan; 26Suppl. 1: 124–30
Rice KM, Savidge GF. NovoSeven® (recombinant factor VIIa) in central nervous system bleeds. Haemostasis 1996 Jan; 26Suppl. 1: 131–4
Scharrer I. Recombinant factor VIIa for patients with inhibitors to factor VIII or IX or factor VII deficiency. German Novoseven Study Group. Haemophilia 1999 Jul; 5(4): 253–9
Lusher J, Ingerslev J, Roberts H, et al. Clinical experience with recombinant factor VIIa. Blood Coagul Fibrinolysis 1998; 9(2): 119–28
Ingerslev J, Freidman D, Gastineau D, et al. Major surgery in haemophilic patients with inhibitors using recombinant factor VIIa. Haemostasis 1996 Jan; 26 Suppl. 1: 118–23
Ingerslev J. Efficacy and safety of recombinant factor VIIa in the prophylaxis of bleeding in various surgical procedures in hemophilic patients with factor VIII and factor IX inhibitors. Semin Thromb Hemost 2000; 26(4): 425–32
Rodriguez-Merchan EC, Wiedel JD, Wallny T, et al. Elective orthopedic surgery for hemophilia patients with inhibitors: new opportunities. Semin Hematol 2004 Jan; 41(1 Suppl. 1): 109–16
Lusher JM. Acute hemarthroses: the benefits of early versus late treatment with recombinant activated factor VII. Blood Coagul Fibrinolysis 2000 Apr; 11Suppl. 1: S45–9
Lusher JM. Recombinant activated factor VII for treatment of intramuscular haemorrhages: a comparison of early versus late treatment. Blood Coagul Fibrinolysis 1998; 9Suppl. 1: S111–4
Parameswaran R, Shapiro AD, Gill JC, et al. Dose effect and efficacy of rFVIIa in the treatment of haemophilia patients with inhibitors: analysis from the Hemophilia and Thrombosis Research Society Registry. Haemophilia 2005; 11(2): 100–6
Kenet G, Lubetsky A, Luboshitz J, et al. A new approach to treatment of bleeding episodes in young hemophilia patients: a single bolus megadose of recombinant activated factor VII (NovoSeven®). J Thromb Haemost 2003 Mar; 1(3): 450–5
Obergfell A, Auvinen MK, Mathew P. Recombinant activated factor VII for haemophilia patients with inhibitors undergoing orthopaedic surgery: a review of the literature. Haemophilia Epub 2007 Dec 12
Sumner MJ, Geldziler BD, Pedersen M, et al. Treatment of acquired haemophilia with recombinant activated FVII: a critical appraisal. Haemophilia 2007; 13(5): 451–61
Hay CRM, Negrier C, Ludlam CA. The treatment of bleeding in acquired haemophilia with recombinant factor VIIa: a multicentre study. Thromb Haemost 1997; 78: 1463–7
Baudo F, De Cataldo F, Gaidano G. Treatment of acquired factor VIII inhibitor with recombinant activated factor VIIa: data from the Italian registry of acquired hemophilia. Haematologica 2004 Jun; 89(6): 759–61
Mariani G, Konkle BA, Ingerslev J. Congenital factor VII deficiency: therapy with recombinant activated factor VII — a critical appraisal. Haemophilia 2006 Jan; 12(1): 19–27
Brenner B, Wiis J. Experience with recombinant-activated factor VII in 30 patients with congenital factor VII deficiency. Hematology 2007 Feb; 12(1): 55–62
Poon MC, Demers C, Jobin F, et al. Recombinant factor VIIa is effective for bleeding and surgery in patients with Glanzmann thrombasthenia. Blood 1999 Dec 1; 94(11): 3951–3
Almeida AM, Khair K, Hann I, et al. The use of recombinant factor VIIa in children with inherited platelet function disorders. Br J Haematol 2003 May; 121(3): 477–81
Abshire T, Kenet G. Recombinant factor VIIa: review of efficacy, dosing regimens and safety in patients with congenital and acquired factor VIII or IX inhibitors. J Thromb Haemost 2004 Jun; 2(6): 899–909
Roberts HR, Monroe DM, White GC. The use of recombinant factor VIIa in the treatment of bleeding disorders. Blood 2004 Dec 15; 104(13): 3858–64
O’Connell KA, Wood JJ, Wise RP, et al. Thromboembolic adverse events after use of recombinant human coagulation factor VIIa. JAMA 2006 Jan 18; 295(3): 293–8
Aledort LM. Comparative thrombotic event incidence after infusion of recombinant factor VIIa versus factor VIII inhibitor bypass activity. J Thromb Haemost 2004 Oct; 2(10): 1700–8
Makris M, Van Veen JJ. Comparative thrombotic event incidence after infusion of recombinant factor VIIa versus factor VIII inhibitor bypass activity — a rebuttal [letter]. J Thromb Haemost 2005; 3(4): 818–9
Sallah S, Isaksen M, Seremetis S, et al. Comparative thrombotic event incidence after infusion of recombinant factor VIIa vs. factor VIII inhibitor bypass activity — a rebuttal [letter]. J Thromb Haemost 2005; 3(4): 820–2
Aledort LM. Comparative thrombotic event incidence after infusion of recombinant factor VIIa vs. factor VIII inhibitor bypass activity — reply to a rebuttal [letter]. J Thromb Haemost 2005; 3(4): 822
Lyseng-Williamson KA, Plosker GL. Recombinant factor VIIa (eptacog alfa): a pharmacoeconomic review of its use in haemophilia in patients with inhibitors to clotting factors VIII or IX. Pharmacoeconomics 2007; 25(12): 1007–29
Ekert H, Brewin T, Boey W, et al. Cost-utility analysis of recombinant factor VIIa (NovoSeven®) in six children with long-standing inhibitors to factor VIII or IX. Haemophilia 2001 May; 7(3): 279–85
Knight C, Paisley S, Wight J, et al. Economic modelling of different treatment strategies for haemophilia A with high-responding inhibitors. Haemophilia 2003 Jul; 9(4): 521–40
Ballal R, Joshi AV, Stephens JM. Lifetime economic value of orthopedic surgery with recombinant activated factor VII in hemophilia patients with inhibitors [abstract no. PHM6]. Value Health 2007 May–Jun; 10(3): A154. Plus poster presented at the 12th Annual Congress of the International Society for Pharmacoeconomics and Outcome Research; 2007 May 19–23; Arlington (VA)
Botteman MF, Ballal R, Joshi A. Economic benefits of knee surgery with recombinant activated factor VII in hemophilia patients with inhibitors [abstract no. P-M-171 plus poster]. 21st Congress of the International Society on Thrombosis and Haemostasis; 2007 Jul 6–12; Geneva
Green C, De Cock E. Cost-consequences of orthopaedic surgery in haemophilia patients with inhibitors [abstract no. POD4]. Value Health 2001 Nov–Dec; 4 (6): 467. Plus poster presented at the 4th Annual European Conference of the International Society for Pharmacoeconomics and Outcomes Research; 2001 Nov 11–13; Cannes
Morfini M, Haya S, Tagariello G, et al. European study on orthopaedic status of haemophilia patients with inhibitors. Haemophilia 2007 Sep; 13(5): 606–12
Medical and Scientific Advisory Council (MASAC) of the National Hemophilia Foundation. Guidelines for emergency department management of individuals with hemophilia [online]. Available from URL: http://www.hemophilia.org/NHFWeb/Resource/StaticPages/menu0/menu5/menu57/175.pdf [Accessed 2008 Feb 22]
Turecek PL, Váradi K, Gritsch H, et al. FEIBA: mode of action. Haemophilia 2004 Sep; 10Suppl. 2: 3–9
Hay CRM, Baglin TP, Collins PW, et al. The diagnosis and management of factor VIII and IX inhibitors: a guideline from the UK Haemophilia Centre Doctors’ Organization (UKHCDO). Br J Haematol 2000 Oct; 111(1): 78–90
Teitel J, Berntorp E, Collins P, et al. A systematic approach to controlling problem bleeds in patients with severe congenital haemophilia A and high-titre inhibitors. Haemophilia 2007; 13: 256–63
Tengborn L, Ingerslev J, Mäkipernaa A, et al. Acquired haemophilia: Nordic guidelines for diagnosis and treatment. Working group on acquired haemophilia of the Nordic haemophilia centres [online]. Available from URL: http://www.medfak.lu.se/pdf/medkirortpdf/koagulation/nordic_guidelines_20070213.pdf [Accessed 2008 Feb 12]
Page MJ, Key NS, Rockwood T. Patient/caregiver assessment of convenience in the use of recombinant activated factor VII (rVIIa; NovoSeven®) in home therapy. Blood Coagul Fibrinolysis 2000 Apr; 11Suppl. 1: S51–2
Bysted BV, Scharling B, Moller T, et al. A randomized, double-blind trial demonstrating bioequivalence of the current recombinant activated factor VII formulation and a new robust 25°C stable formulation. Haemophilia 2007; 13(5): 527–32
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Croom, K.F., McCormack, P.L. Recombinant Factor VIIa (Eptacog Alfa). BioDrugs 22, 121–136 (2008). https://doi.org/10.2165/00063030-200822020-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00063030-200822020-00005