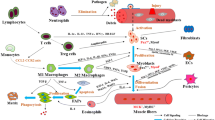
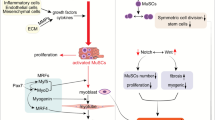
Abstract
Skeletal muscles are heterogeneous tissue containing different types of muscle fibers. Their distribution depends on heredity, type of exercise, sex, age, and muscle type. In addition, stem cells (myosatellitocytes) are found in large amounts in the muscle tissue. Myosatellitocytes are the main material for regeneration of microtears of muscle fibers always occuring during intensive physical exercises. Myosatellitocytes are capable of long-term storage in an inactive “dormant” state, but they can be rapidly activated to provide an efficient repair of damaged muscle fibers. The metabolism of myosatellitocytes and myoblasts and their migration into the damaged area are regulated by a complex system of cytokines and transcription factors, the activity of which depends on many factors. Microtears initiating the development of the inflammatory process and activation of myosatellitocytes is a determining factor. The study into molecular mechanisms of the relationship between inflammatory processes in muscle tissue and changes in myosatellitocyte metabolism is of fundamental importance and is necessary for the selection of efficient methods for muscle tissue recovery.

Similar content being viewed by others
REFERENCES
Abreu, P., Serna, J.D.C., Munhoz, A.C., and Kowaltowski, A.J., Calorie restriction changes muscle satellite cell proliferation in a manner independent of metabolic modulation, Mech. Ageing Dev., 2020, vol. 192, art. ID 111362. https://doi.org/10.1016/j.mad.2020.111362
Ahmetov, I.I., Druzhevskaya, A., Lyubaeva, E.V., Popov, D.V., Vinogradova, O.L., Williams, A.G., The dependence of preferred competitive racing distance on muscle fibre composition and ACTN3 genotype in speed skaters, Exp. Physiol., 2011, vol. 96, no. 12, pp. 1302–1310. https://doi.org/10.1113/expphysiol.2011.060293
Anderson, J.E., A role for nitric oxide in muscle repair: nitric oxide-mediated activation of muscle satellite cells, Mol. Biol. Cell., 2000, vol. 11, no. 5, pp. 1859–1874. https://doi.org/10.1091/mbc.11.5.1859
Bisetto, S., Wright, M.C., Nowak, R.A., Lepore, A.C., Khurana, T.S., Loro, E., and Philp, N.J., New insights into the lactate shuttle: role of MCT4 in the modulation of the exercise capacity, Science, 2019, vol. 22, pp. 507–518. https://doi.org/10.1016/j.isci.2019.11.041
Blondelle, J., Shapiro, P., Domenighetti, A.A., and Lange, S., Cullin E3 ligase activity is required for myoblast differentiation, J. Mol. Biol., 2017, vol. 429, no. 7, pp. 1045–1066. https://doi.org/10.1016/j.jmb.2017.02.012
Bosch-Presegue, L. and Vaquero, A., Sirtuin-dependent epigenetic regulation in the maintenance of genome integrity, FEBS J., 2015, vol. 282, no. 9, pp. 1745–1767. https://doi.org/10.1111/febs.13053
Brack, A.S., Conboy, M.J., Roy, S., Lee, M., Kuo, C.J., Keller, C., and Rando, T.A., Increased Wnt signaling during aging alters muscle stem cell fate and increases fibrosis, Science, 2007, vol. 317, no. 5839, pp. 807–810. https://doi.org/10.1126/science.1144090
Brack, A.S. and Rando, T.A., Tissue-specific stem cells: lessons from the skeletal muscle satellite cell, Cell Stem Cell, 2012, vol. 10, no. 5, pp. 504–514. https://doi.org/10.1016/j.stem
Britto, F.A., Gnimassou, O., De Groote, E., Balan, E., Warnier, G., Everard, A., Cani, P.D., and Deldicque, L., Acute environmental hypoxia potentiates satellite cell-dependent myogenesis in response to resistance exercise through the inflammation pathway in human, FASEB J., 2020, vol. 34, no, 1, pp. 1885–1900. https://doi.org/10.1096/fj.201902244R
Canto, C. and Auwerx, J., Calorie restriction: is AMPK a key sensor and effector?, Physiology, 2011, vol. 26, pp. 214–224. https://doi.org/10.1152/physiol.00010.2011
Cerletti, M., Jang, Y.C., Finley, L.W., Haigis, M.C., and Wagers, A.J., Short-term calorie restriction enhances skeletal muscle stem cell function, Cell Stem Cell, 2012, vol. 10, no. 5, pp. 515–519. https://doi.org/10.1016/j.stem.2012.04.002
Collins-Hooper, H., Woolley, T.E., Dyson, L., Patel, A., Potter, P., Baker, R.E., Gaffney, E.A., Maini, P.K., Dash, P.R., and Patel, K., Age-related changes in speed and mechanism of adult skeletal muscle stem cell migration, Stem Cells, 2012, vol. 30, no. 6, pp. 1182–1195. https://doi.org/10.1002/stem.1088
Dell’Orso, S., Juan, A.H., Ko, K.D., Naz, F., Perovanovic, J., Gutierrez-Cruz, G., Feng, X., and Sartorelli, V., Single cell analysis of adult mouse skeletal muscle stem cells in homeostatic and regenerative conditions, Development, 2019, vol. 146, no. 12, art. ID dev174177. https://doi.org/10.1242/dev.174177
Dong, Z., Saikumar, P., Weinberg, J.M., and Venkatachalam, M.A., Calcium in cell injury and death, Annu. Rev. Pathol., 2006, vol. 1, pp. 405–434. https://doi.org/10.1146/annurev.pathol.1.110304.100218
Fang, Y., Tang, S., and Li, X., Sirtuins in metabolic and epigenetic regulation of stem cells, Trends Endocrinol. Metab., 2019, vol. 30, pp. 177–188. https://doi.org/10.1016/j.tem.2018.12.002
Fuchs, E. and Chen, T., A matter of life and death: self-renewal in stem cells, EMBO Rep., 2013, vol. 14, no. 1, pp. 39–48. https://doi.org/10.1038/embor.2012.197
Fukada, S., Uezumi, A., Ikemoto, M., Masuda, S., Segawa, M., Tanimura, N., Yamamoto, H., Miyagoe-Suzuki, Y. and Takeda, S., Molecular signature of quiescent satellite cells in adult skeletal muscle, Stem Cells, 2007, vol. 25, no. 10, pp. 2448–2459. https://doi.org/10.1634/stemcells.2007-0019
Fuku, N., Kumagai, H., and Ahmetov, I., Genetics of muscle fiber composition, in Sports, Exercise, and Nutritional Genomics, 2019, vol. 14, pp. 295–314. https://doi.org/10.1016/B978-0-12-816193-7.00014-2
Gerrits, M.F., Ghosh, S., Kavaslar, N., Hill, B., Tour, A., Seifert, E.L., Beauchamp, B., Gorman, S., Stuart, J., Dent, R., McPherson, R., and Harper, M.E., Distinct skeletal muscle fiber characteristics and gene expression in diet-sensitive versus diet-resistant obesity, J. Lipid Res., 2010, vol. 51, no. 8, pp. 2394–2404. https://doi.org/10.1194/jlr.P005298
Haizlip, K.M., Harrison, B.C., and Leinwand, L.A., Sex-based differences in skeletal muscle kinetics and fiber-type composition, Physiology, 2015, vol. 30, no. 1, pp. 30–39. https://doi.org/10.1152/physiol.00024.2014
Halestrap, A.P. and Wilson, M.C., The monocarboxylate transporter family—Role and regulation, IUBMB Life, 2012, vol. 64, no. 2, pp. 109–119. https://doi.org/10.1002/iub.572
Hardie, D.G., Ross, F.A., and Hawley, S.A., AMPK: a nutrient and energy sensor that maintains energy homeostasis, Nat. Rev. Mol. Cell. Biol., 2012, vol. 13, no. 4, pp. 251–262. https://doi.org/10.1038/nrm3311
Hendrickse, P.W., Venckunas, T., Platkevicius, J., Kairaitis, R., Kamandulis, S., Snieckus, A., Stasiulis, A., Vitkiene, J., Subocius, A., and Degens, H., Endurance training-induced increase in muscle oxidative capacity without loss of muscle mass in younger and older resistance-trained men, Eur. J. Appl. Physiol., 2021, vol. 121, no. 11, pp. 3161–3172. https://doi.org/10.1007/s00421-021-04768-4
Jing, H. and Lin, H., Sirtuins in epigenetic regulation, Chem. Rev., 2015, vol. 115, no. 6, pp. 2350–2375. https://doi.org/10.1021/cr500457h
Kantarci, A. and Van Dyke, T.E., Lipoxins in chronic inflammation, Crit. Rev. Oral Biol. Med., 2003, vol. 14, no. 1, pp. 4–12. https://doi.org/10.1177/154411130301400102
Karalaki, M., Fili, S., Philippou, A. and Koutsilieris, M., Muscle regeneration: cellular and molecular events, In Vivo, 2009, vol. 23, no. 5, pp. 779–796.
Klein, C.S., Marsh, G.D., Petrella, R.J., and Rice, C.L., Muscle fiber number in the biceps brachii muscle of young and old men, Muscle Nerve, 2003, vol. 28, no. 1, pp. 62–68. https://doi.org/10.1002/mus.10386
Kumagai, H., Tobina, T., Ichinoseki-Sekine, N., Kakigi, R., Tsuzuki, T., Zempo, H., Shiose, K., Yoshimura, E., Kumahara, H., Ayabe, M., Higaki, Y., Yamada, R., Kobayashi, H., Kiyonaga, A., Naito, H., Tanaka, H., Fuku, N., Role of selected polymorphisms in determining muscle fiber composition in Japanese men and women, J. Appl. Physiol., 2018, vol. 124, no. 5, pp. 1377–1384. https://doi.org/10.1152/japplphysiol.00953.2017
Kumar, V., Abbas, A.K. and Aster, J.C., Robbins Basic Pathology, Elsevier, 2012.
Kutseryb, T., Hrynkiv, M., Vovkanych, L., and Muzyka, F., Influence of basketball training on the features of women’s physique, J. Phys. Educ. Sport, 2019, vol. 19, no. 4, pp. 2384–2389. https://doi.org/10.7752/jpes.2019.04361
Lamont, L.A., Tranquilli, W.J., and Grimm, K.A., Physiology of pain, Vet. Clin. North Am. Small Anim. Pract., 2000, vol. 30, no. 4, pp. 703–728. https://doi.org/10.1016/s0195-5616(08)70003-2
Latil, M., Rocheteau, P., Chatre, L., Sanulli, S., Mémet, S., Ricchetti, M., Tajbakhsh, S., and Chrétien, F., Skeletal muscle stem cells adopt a dormant cell state post mortem and retain regenerative capacity, Nat. Commun., 2012, vol. 3, art. ID 903. https://doi.org/10.1038/ncomms1890
Le Moal, E., Pialoux, V., Juban, G., Groussard, C., Zouhal, H., Chazaud, B., and Mounier, R., Redox control of skeletal muscle regeneration, Antioxid. Redox Signal, 2017, vol. 27, no. 5, 276–310. https://doi.org/10.1089/ars.2016.6782
Liu L, Cheung, T.H., Charville, G.W., Hurgo, B.M., Leavitt, T., Shih, J., Brunet, A., and Rando, T.A., Chromatin modifications as determinants of muscle stem cell quiescence and chronological aging, Cell Rep., 2013, vol. 4, no. 1, pp. 189–204. https://doi.org/10.1016/j.celrep.2013.05.043
Marzetti, E., Lawler, J.M., Hiona, A., Manini, T., Seo, A.Y., and Leeuwenburgh, C., Modulation of age-induced apoptotic signaling and cellular remodeling by exercise and calorie restriction in skeletal muscle, Free Radical Biol. Med., 2008, vol. 44, no. 2, pp. 160–168. https://doi.org/10.1016/j.freeradbiomed.2007.05.028
Mauro, A., Satellite cell of skeletal muscle fibers, J. Biophys. Biochem. Cytol., 1961, vol. 9, no. 2, pp. 493–495. https://doi.org/10.1083/jcb.9.2.493
Mayer, U., Integrins: redundant or important players in skeletal muscle?, J. Biol. Chem., 2003, vol. 278, no. 17, pp. 14587–14590. https://doi.org/10.1074/jbc.R200022200
May-Simera, H.L. and Kelley, M.W., Cilia, Wnt signaling, and the cytoskeleton, Cilia, 2012, vol. 1, no. 1, art. ID 7. https://doi.org/10.1186/2046-2530-1-7
Moussaieff, A., Rouleau, M., Kitsberg, D., Cohen, M., Levy, G., Barasch, D., Nemirovski, A., Shen-Orr, S., Laevsky, I., Amit, M., Bomze, D., Elena-Herrmann, B., Scherf, T., Nissim-Rafinia, M., Kempa, S., Itskovitz-Eldor, J., Meshorer, E., Aberdam, D., and Nahmias, Y., Glycolysis-mediated changes in acetyl-CoA and histone acetylation control the early differentiation of embryonic stem cells, Cell Metab., 2015, vol. 21, no. 3, pp. 392–402. https://doi.org/10.1016/j.cmet.2015.02.002
Nalbandian, M., Radak, Z., and Takeda, M., Lactate metabolism and satellite cell fate, Front. Physiol., 2020, vol. 11, art. ID 610983. https://doi.org/10.3389/fphys.2020.610983
Nathan, C. and Cunningham-Bussel, A., Beyond oxidative stress: an immunologist’s guide to reactive oxygen species, Nat. Rev. Immunol., 2013, vol. 13, pp. 349–361. https://doi.org/10.1038/nri3423
Needleman, P., Turk, J., Jakschik, B.A., Morrison, A.R., and Lefkowith, J.B., Arachidonic acid metabolism, Annu. Rev. Biochem., 1986, vol. 55, pp. 69–102. https://doi.org/10.1146/annurev.bi.55.070186.000441
Oishi, Y., Tsukamoto, H., Yokokawa, T., Hirotsu, K., Shimazu, M., Uchida, K., Tomi, H., Higashida, K., Iwanaka, N., and Hashimoto, T., Mixed lactate and caffeine compound increases satellite cell activity and anabolic signals for muscle hypertrophy, J. Appl. Physiol., 2015, vol. 118, no. 6, pp. 742–749. https://doi.org/10.1152/japplphysiol.00054.2014
Otto, A., Collins-Hooper, H., Patel, A., Dash, P.R., and Patel, K., Adult skeletal muscle stem cell migration is mediated by a blebbing/amoeboid mechanism, Rejuvenation Res., 2011, vol. 14, no. 3, pp. 249–260. https://doi.org/10.1089/rej.2010.1151
Pallafacchina, G., Blaauw, B., and Schiaffino, S., Role of satellite cells in muscle growth and maintenance of muscle mass, Nutr., Metab. Cardiovasc. Dis., 2013, vol. 23, pp. 12–18. https://doi.org/10.1016/j.numecd2012.02.002D
Kaipainen, A., Greene, E.R., and Huang, S., Cytochrome P450-derived eicosanoids: the neglected pathway in cancer, Cancer Metastasis Rev., 2010, vol. 29, no. 4, pp. 723–735. https://doi.org/10.1007/s10555-010-9264-x
Quintero, A.J., Wright, V.J., Fu, F.H., and Huard, J., Stem cells for the treatment of skeletal muscle injury, Clin. Sports Med., 2009, vol. 28, no. 1, pp. 1–11. https://doi.org/10.1016/j.csm.2008.08.009
Radmark, O., Werz, O., Steinhilber, D., and Samuelsson, B., 5-Lipoxygenase, a key enzyme for leukotriene biosynthesis in health and disease, Biochim. Biophys. Acta, 2015, vol. 1851, no. 4, pp. 331–339. https://doi.org/10.1016/j.bbalip.2014.08.012
Ricciotti, E. and FitzGerald, G.A., Prostaglandins and inflammation, Arterioscler., Thromb., Vasc. Biol., 2011, vol. 31, no. 5, pp. 986–1000. https://doi.org/10.1161/ATVBAHA.110.207449
Rocheteau, P., Vinet, M., and Chretien, F., Dormancy and quiescence of skeletal muscle stem cells, Results Probl. Cell Differ., 2015, vol. 56, pp. 215–235. https://doi.org/10.1007/978-3-662-44608-9_10
Ryall, J.G., Metabolic reprogramming as a novel regulator of skeletal muscle development and regeneration, FEBS J., 2013, vol. 280, pp. 4004–4013. https://doi.org/10.1111/febs.12189
Schmidt, M., Schüler, S.C., Hüttner, S.S., von Eyss, B., and von Maltzahn, J., Adult stem cells at work: regenerating skeletal muscle, Cell Mol. Life Sci., 2019, vol. 76, no. 13, pp. 2559–2570. https://doi.org/10.1007/s00018-019-03093-6
Schoenfeld, B.J., The mechanisms of muscle hypertrophy and their application to resistance training, J. Strength Cond. Res., 2010, vol. 24, no. 10, pp. 2857–2872. https://doi.org/10.1519/JSC.0b013e3181e840f3
Scott, W., Stevens, J., and Binder-Macleod, S.A., Human skeletal muscle fiber type classifications, Phys. Ther., 2001, vol. 81, no. 11, pp. 1810–1816. PMID: 11694174
Suwa, M., Nakamura, T., Katsuta, S., Heredity of muscle fiber composition and correlated response of the synergistic muscle in rats, Am. J. Physiol., 1996, vol. 271, no. 2, pp. R432–R436. https://doi.org/10.1152/ajpregu.1996.271.2.R432
Sybil, M.G., Pervachuk, R.V., and Trach, V.M., Personalization of freestyle wrestlers’ training process by influence the anaerobic systems of energy supply, J. Phys. Educ. Sport., 2015, vol. 15, no. 2, pp. 225–228. https://doi.org/10.7752/jpes.2015.02035
Tang, A.H. and Rando, T.A., Induction of autophagy supports the bioenergetic demands of quiescent muscle stem cell activation, EMBO J., 2014, vol. 33, no. 23, pp. 2782–2797. https://doi.org/10.15252/embj.201488278
Theret, M., Gsaier, L., Schaffer, B., Juban, G., Ben Larbi, S., Weiss-Gayet, M., Bultot, L., Collodet, C., Foretz, M., Desplanches, D., Sanz, P., Zang, Z., Yang, L., Vial, G., Viollet, B., Sakamoto, K., Brunet, A., Chazaud, B., and Mounier, R., AMPKα1-LDH pathway regulates muscle stem cell self-renewal by controlling metabolic homeostasis, EMBO J., 2017, vol. 36, no. 13, pp. 1946–1962. https://doi.org/10.15252/embj.201695273
Vierck, J., O’Reilly, B., Hossner, K., Antonio, J., Byrne, K., Bucci, L., and Dodson, M., Satellite cell regulation following myotrauma caused by resistance exercise, Cell Biol. Int., 2000, vol. 24, no. 5, 263–272. https://doi.org/10.1006/cbir.2000.0499
Wilkinson, D.J., Piasecki, M., and Atherton, P.J., The age-related loss of skeletal muscle mass and function: Measurement and physiology of muscle fibre atrophy and muscle fibre loss in humans, Ageing Res. Rev., 2018, vol. 47, pp. 123–132. https://doi.org/10.1016/j.arr.2018.07.005
Willkomm, L., Schubert, S., Jung, R., Elsen, M., Borde, J., Gehlert, S., Suhr, F., and Bloch, W., Lactate regulates myogenesis in C2C12 myoblasts in vitro, Stem Cell Res., 2014, vol. 12, no. 3, pp. 742–753. https://doi.org/10.1016/j.scr.2014.03.004
Wright, E.M. and Woodson, J.F., Clinical assessment of pain in laboratory animals, in Rollin, B.E., Kesel, M.L., Eds., The Experimental Animal in Biologic Research, Boca Raton: CRC Press, 1990, pp. 205–216.
Yamakawa, H., Kusumoto, D., Hashimoto, H., and Yuasa, S., Stem cell aging in skeletal muscle regeneration and disease, Int. J. Mol. Sci., 2020, vol. 21, no. 5, art. ID 1830. https://doi.org/10.3390/ijms21051830
Zammit, P.S., Relaix, F., Nagata, Y., Ruiz, A.P., Collins, C.A., Partridge, T.A., Beauchamp, J.R., Pax7 and myogenic progression in skeletal muscle satellite cells, J. Cell Sci., 2006, vol. 119, pp. 1824–1832. https://doi.org/10.1242/jcs.02908
Funding
This study was not funded by any specific grant from the financial institutions in the state, commercial or noncommercial sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest. This article contains no studies involving human participants and animals as objects of study.
Additional information
Translated by A. Barkhash
About this article
Cite this article
Hashchyshyn, V., Tymochko-Voloshyn, R., Paraniak, N. et al. Regeneration of Skeletal Muscle Fibers and Regulation of Myosatellitocytes Metabolism. Cytol. Genet. 56, 253–260 (2022). https://doi.org/10.3103/S0095452722030033
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S0095452722030033