Abstract
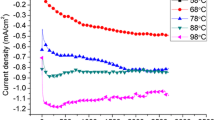
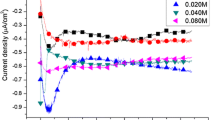
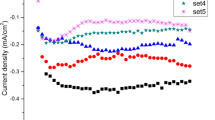
The effects of pH value on CdS thin films fabricated by using electrodeposition were investigated in detail. The pH values of the final solutions were adjusted between 1 and 5. Aqueous solutions of HCl and NaOH were used to adjust the pH. The reaction rates were changed by the pH due to the fact that the pH affected the rate of release of sulphide from sodium thiosulfate. The structural, optical and morphological properties of the films were analyzed by using X-ray diffraction, Ultraviolet-visible spectroscopy and scanning electron microscopy, respectively. Compact films with good crystallinity were obtained at pH value of 4 and 5.
Similar content being viewed by others
References
M. N. Mammadov, A. ShAliyev and M. Elrouby, Int. J. Thin Film Sci. Tech. 1, 43 (2012).
M. Fathy, A. El-Hady, B. Kashyout, S. Elyamny, G. D. Roston and A. A. Bishara, Int. J. Electrochem. Sci. 9, 6155 (2014).
M. Takahashi, S. Hasegawa, M. Watanabe, T. Miyuki, S. Ikeda and K. Iida, Int. J. Appl. Chem. 32, 359 (2002).
D. Lincot, Thin Solid Films 487, 40 (2005).
E. O. Okechukwu and D. N. Okoli, J. Mater. Sci. Appl. 1, 282 (2015).
G. Sasikala, R. Dhanasekaran and C. Subramanian, Thin Solid Films 302, 71 (1997).
R. N. Bhowmik, M. NrisimhaMurty and E. SekharSrinadhu, PMC Phys. B 1, 20 (2008).
W. Y. Gan, K. Chiang, M. Brungs and R. Amal, Int. J. Nanotechnol. 4, 574 (2007).
Y. Hu, D. Ma, X. Chen, Y. Chen and J. Chen, Int. J. Surf. Sci. and Engin. 8, 28 (2014).
Y. Kim and J-Y. Leem, J. Korean Phys. Soc. 67, 870 (2015).
H. Ahn and Y. Um, J. Korean Phys. Soc. 64, 1600 (2014).
H. Y. R. Atapattu, D. S. M. De Silva, K. A. S. Pathiratne and I. M. Dharmadasa, J. Mater. Sci: Mater. Electron. 27, 5415 (2016).
F. Kadrgan, D. Mao, W. Song, T. Ohnoe and B. Mc-Candless, Turk. J. Chem. 24, 21 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Altiokka, B., Yildirim, A.K. Electrodeposition of CdS Thin Films at Various pH Values. J. Korean Phys. Soc. 72, 687–691 (2018). https://doi.org/10.3938/jkps.72.687
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3938/jkps.72.687